FOLFIRI
Common complications6
More than 10 in every 100 people have one or more of the side effects listed below.
- An increased risk of getting an infection from a drop in white blood cells – it is harder to fight infections and you can become very ill. You may have headaches, aching muscles, a cough, a sore throat, pain passing urine, or you may feel cold and shivery. If you have a severe infection this can be life-threatening. Contact your treatment centre straight away if you have any of these effects or if your temperature goes above 38 °C. You will have regular blood tests to check your blood cell levels
- Tiredness and breathlessness due to a drop in red blood cells (anaemia) – you may need a blood transfusion
- Bruising more easily due to a drop in platelets – you may have nosebleeds or bleeding gums after brushing your teeth. Or you may have lots of tiny red spots or bruises on your arms or legs (known as petechia)
- Tiredness and weakness (fatigue) during and after treatment – most people find their energy levels are back to normal within 6 months to a year
- Numbness or tingling in the fingers and toes happens to nearly everyone having oxaliplatin and is usually worse if you are cold. You may have trouble with fiddly tasks such as doing up buttons. This can start a few days or weeks after treatment and usually goes away within a few months of the treatment finishing
- Feeling of nausea happens to about 7 out of every 10 people (70%) but is usually well controlled with anti-nausea drugs. If the nausea is not controlled, let your nurse know as there are other anti-nausea medicines that may work better for you
- Pain in the vein during the infusion of oxaliplatin or folinic acid – tell your nurse if this happens. Sometimes the drugs may need to be given more slowly
- Some people develop soreness, redness and peeling on the palms of the hands and soles of the feet (plantar palmar syndrome). This may cause tingling, numbness, pain and dryness
- Diarrhea happens to 6 out of 10 people (60%) – tell your doctor or nurse if it becomes severe, if you can’t drink to replace the lost fluid, or if it carries on for more than 3 days.Your nurse may give you anti diarrhea medicine to take home with you after chemotherapy
- A sore mouth (stomatitis) happens to 4 out of 10 people (40%)
- Women may stop having periods (amenorrhoea) but this may be temporary
- Loss of fertility – you may not be able to become pregnant or father a child after this treatment. Talk to your doctor before starting treatment if you think you may want to have a baby in the future. Men may be able to store sperm before starting treatment
Экспозиция к лечению и лечение второй линии
Было отмечено существенное различие между двумя ветвями лечения относительно продолжительности лечения: 3,5 месяца в ветви FOLFIFI плюс цетуксимаб по сравнению с 5,2 месяца в ветви FOLFIRI плюс бевацизумаб. Подобно этому, число циклов, в которых была снижена доза химиотерапевтических препаратов, было выше в ветви цетуксимаба (27%), чем в ветви бевацизумаба (15%) (Р
Таблица 1. Назначение лечения и лечение второй линии
| Параметр | FOLFIRI + цетуксимаб | FOLFIRI + бевацизумаб | Итог |
|---|---|---|---|
| Пациенты (N) | 41 | 46 | 87 |
| Продолжительность терапии, месяцы | |||
| Медиана (колебания) | 3,5 (0,03–11,3) | 5,2 (0,03–14,4) | 4,7 (0,03–14,4) |
| Отсутствующий, n | 3 | 2 | 5 |
| Циклы лечения на одного пациента | |||
| Медиана (колебания) | 7 (1–24) | 12 (1–47) | 9 (1–47) |
| Подлежащие оценке циклы лечения | N (%)325 (100) | N (%)553 (100) | Показатель ра |
| Циклы со снижением дозыb | 89 (27) | 80 (15) | |
| Циклы с отсроченным лечениемc | 34 (11) | 69 (13) | 0,387 |
| Лечение второй линии | N (%) | N (%) | N (%) |
| Пациенты, получившие лечение 5-ФУ, иринотеканом и оксалиплатином, “все три препарата” | 27 (54) | 29 (63) | 56 (58) |
| Пациенты, получившие лечение цетуксимабом и бевацизумабом, “оба антитела” | 25 (50) | 5 (11) | 30 (31) |
| Пациенты, получившие лечение 5-ФУ, иринотеканом и оксалиплатином, цетуксимабом и бевацизумабом, “все пять препаратов” | 20 (40) | 5 (11) | 25 (26) |
| Бевацизумаб после прогрессирования | – | 15 (33) | – |
аБыло определено как bТест точности Фишера. сБыло определено как циклы лечения, отсроченные >16 дней. FOLFIRI — инфузионные 5-фторурацил, фолиновая кислота, иринотекан. 5-ФУ — 5-фторурацил. |
Число пациентов, которые получили все три активных химиотерапевтических препарата (5-ФУ/капецитабин, иринотекан и оксалиплатин) на протяжении курса лечения мКРР, было сравнимым между ветвями А и В (54% по сравнению с 63%; таблица 1). Однако число пациентов, которые получили оба антитела (цетуксимаб и бевацизумаб), существенно различалось между двумя ветвями лечения (50% пациентов ветви А получали бевацизумаб и 11% пациентов в ветви В получали цетуксимаб на протяжении всего курса лечения; Р
FOLFOX4edit
Adjuvant treatment in patients with stage III colon cancer is recommended for 12 cycles, every 2 weeks. The recommended dose schedule given every two weeks is as follows:
Day 1: Oxaliplatin 85 mg/m2 IV infusion in 250-500 mL D5W and leucovorin 200 mg/m2 IV infusion in D5W both given over 120 minutes at the same time in separate bags using a Y-line, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
Day 2: Leucovorin 200 mg/m2 IV infusion over 120 minutes, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
FOLFOX4 regime
| FOLFOX4 | ||||
|---|---|---|---|---|
| drug | dose | administration | time | term |
| Oxaliplatin | 85 mg/m2 | IV infusion | 2 h | day 1 |
| Folinic acid | 200 mg/m2 | IV infusion | 2 h | day 1 + 2 |
| Fluorouracil | 400 mg/m2 | IV bolus | 2 min | day 1 + 2 |
| Fluorouracil | 600 mg/m2 | IV infusion | 22 h | day 1 + 2 |
Premedication with antiemetics, including 5-HT3 blockers with or without dexamethasone, is recommended.
Common complications6
More than 10 in every 100 people have one or more of the side effects listed below.
- An increased risk of getting an infection from a drop in – it is harder to fight infections and you can become very ill. You may have headaches, aching muscles, a cough, a sore throat, pain passing urine, or you may feel cold and shivery. If you have a severe infection this can be life-threatening. Contact your treatment centre straight away if you have any of these effects or if your temperature goes above 38 °C. You will have regular blood tests to check your blood cell levels
- Tiredness and breathlessness due to a drop in red blood cells () – you may need a blood transfusion
- Bruising more easily due to a drop in platelets – you may have nosebleeds or bleeding gums after brushing your teeth. Or you may have lots of tiny red spots or bruises on your arms or legs (known as petechia)
- Tiredness and weakness (fatigue) during and after treatment – most people find their energy levels are back to normal within 6 months to a year
- Numbness or tingling in the fingers and toes happens to nearly everyone having oxaliplatin and is usually worse if you are cold. You may have trouble with fiddly tasks such as doing up buttons. This can start a few days or weeks after treatment and usually goes away within a few months of the treatment finishing
- Feeling of nausea happens to about 7 out of every 10 people (70%) but is usually well controlled with anti-nausea drugs. If the nausea is not controlled, let your nurse know as there are other anti-nausea medicines that may work better for you
- Pain in the vein during the infusion of oxaliplatin or folinic acid – tell your nurse if this happens. Sometimes the drugs may need to be given more slowly
- Some people develop soreness, redness and peeling on the palms of the hands and soles of the feet (). This may cause tingling, numbness, pain and dryness
- happens to 6 out of 10 people (60%) – tell your doctor or nurse if it becomes severe, if you can’t drink to replace the lost fluid, or if it carries on for more than 3 days.Your nurse may give you anti diarrhea medicine to take home with you after chemotherapy
- A sore mouth () happens to 4 out of 10 people (40%)
- Women may stop having periods () but this may be temporary
- Loss of fertility – you may not be able to become pregnant or father a child after this treatment. Talk to your doctor before starting treatment if you think you may want to have a baby in the future. Men may be able to store sperm before starting treatment
FOLFOX4
treatment in patients with stage III colon cancer is recommended for 12 cycles, every 2 weeks. The recommended dose schedule given every two weeks is as follows:
Day 1: Oxaliplatin 85 mg/m2 IV infusion in 250-500 mL and leucovorin 200 mg/m2 IV infusion in D5W both given over 120 minutes at the same time in separate bags using a Y-line, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
Day 2: Leucovorin 200 mg/m2 IV infusion over 120 minutes, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
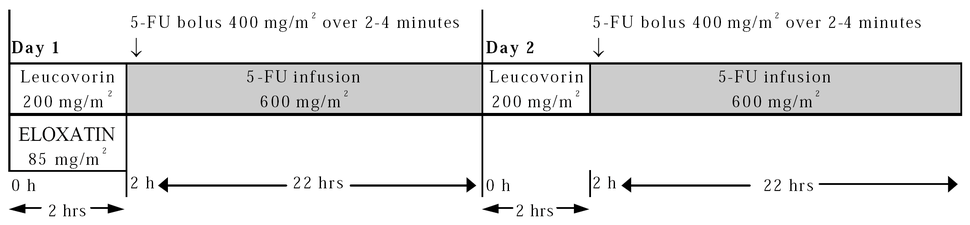
FOLFOX4 regime
| FOLFOX4 | ||||
|---|---|---|---|---|
| drug | dose | administration | time | term |
| 85 mg/m2 | infusion | 2 h | day 1 | |
| 200 mg/m2 | IV infusion | 2 h | day 1 + 2 | |
| 400 mg/m2 | IV bolus | 2 min | day 1 + 2 | |
| Fluorouracil | 600 mg/m2 | IV infusion | 22 h | day 1 + 2 |
Premedication with antiemetics, including 5-HT3 blockers with or without , is recommended.
Administration of FOLFOX5
FOLFOX is given directly into the bloodstream through an intravenous line. It can be given through a thin, short tube (a cannula) put into a vein in the arm each time one has a treatment. It may also be given through a central line, a portacath, or a PICC line. These are long, plastic tubes that give the drugs directly into a large vein in the chest. The tube can stay in place throughout the treatment.
Chemotherapy can be given as cycles of treatment. The number of cycles depends on the situation but may be up to 12. Each treatment cycle lasts 2 weeks.
On the first day oxaliplatin is given through a drip over 2 hours. Folinic acid is given concurrently. This followed by an injection of fluorouracil into the cannula or central line. An infusion of 5FU is then started through a drip or pump, which lasts for 22 hours.
On the second day folinic acid is given as an injection or through a drip for 2 hours. This is followed by an injection of fluorouracil, followed by another fluorouracil infusion through a drip or pump for 22 hours.
Patient then has no treatment for 12 days. After that they start another treatment cycle.
If they have a central line they may be able to have the infusions of fluorouracil at home. If they are at home, they have the infusions through a small pump. They can keep the pump in a small bag, or a bag on a belt (like a bum bag). They’ll need to go back to the hospital for the second day of their treatment, to have the pump changed. Sometimes a chemotherapy nurse may be able to change the infusion at their home. In some countries (like Germany), it is common practice that patients disconnect the infusion pump themselves at home.
When the second infusion of 5FU is finished, the nurse will disconnect the drip and take the cannula out. If they have a central line this will stay in but it will be blocked with a plastic cap until they start your next treatment cycle.
Common complications6edit
More than 10 in every 100 people have one or more of the side effects listed below.
- An increased risk of getting an infection from a drop in white blood cells – it is harder to fight infections and you can become very ill. You may have headaches, aching muscles, a cough, a sore throat, pain passing urine, or you may feel cold and shivery. If you have a severe infection this can be life-threatening. Contact your treatment centre straight away if you have any of these effects or if your temperature goes above 38 °C. You will have regular blood tests to check your blood cell levels
- Tiredness and breathlessness due to a drop in red blood cells (anaemia) – you may need a blood transfusion
- Bruising more easily due to a drop in platelets – you may have nosebleeds or bleeding gums after brushing your teeth. Or you may have lots of tiny red spots or bruises on your arms or legs (known as petechia)
- Tiredness and weakness (fatigue) during and after treatment – most people find their energy levels are back to normal within 6 months to a year
- Numbness or tingling in the fingers and toes happens to nearly everyone having oxaliplatin and is usually worse if you are cold. You may have trouble with fiddly tasks such as doing up buttons. This can start a few days or weeks after treatment and usually goes away within a few months of the treatment finishing
- Feeling of nausea happens to about 7 out of every 10 people (70%) but is usually well controlled with anti-nausea drugs. If the nausea is not controlled, let your nurse know as there are other anti-nausea medicines that may work better for you
- Pain in the vein during the infusion of oxaliplatin or folinic acid – tell your nurse if this happens. Sometimes the drugs may need to be given more slowly
- Some people develop soreness, redness and peeling on the palms of the hands and soles of the feet (plantar palmar syndrome). This may cause tingling, numbness, pain and dryness
- Diarrhea happens to 6 out of 10 people (60%) – tell your doctor or nurse if it becomes severe, if you can’t drink to replace the lost fluid, or if it carries on for more than 3 days.Your nurse may give you anti diarrhea medicine to take home with you after chemotherapy
- A sore mouth (stomatitis) happens to 4 out of 10 people (40%)
- Women may stop having periods (amenorrhoea) but this may be temporary
- Loss of fertility – you may not be able to become pregnant or father a child after this treatment. Talk to your doctor before starting treatment if you think you may want to have a baby in the future. Men may be able to store sperm before starting treatment
FOLFOX4
Adjuvant treatment in patients with stage III colon cancer is recommended for 12 cycles, every 2 weeks. The recommended dose schedule given every two weeks is as follows:
Day 1: Oxaliplatin 85 mg/m2 IV infusion in 250-500 mL D5W and leucovorin 200 mg/m2 IV infusion in D5W both given over 120 minutes at the same time in separate bags using a Y-line, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
Day 2: Leucovorin 200 mg/m2 IV infusion over 120 minutes, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
FOLFOX4 regime
| FOLFOX4 | ||||
|---|---|---|---|---|
| drug | dose | administration | time | term |
| Oxaliplatin | 85 mg/m2 | IV infusion | 2 h | day 1 |
| Folinic acid | 200 mg/m2 | IV infusion | 2 h | day 1 + 2 |
| Fluorouracil | 400 mg/m2 | IV bolus | 2 min | day 1 + 2 |
| Fluorouracil | 600 mg/m2 | IV infusion | 22 h | day 1 + 2 |
Premedication with antiemetics, including 5-HT3 blockers with or without dexamethasone, is recommended.
Administration of FOLFOX5
FOLFOX is given directly into the bloodstream through an intravenous line. It can be given through a thin, short tube (a cannula) put into a vein in the arm each time one has a treatment. It may also be given through a central line, a portacath, or a PICC line. These are long, plastic tubes that give the drugs directly into a large vein in the chest. The tube can stay in place throughout the treatment.
Chemotherapy can be given as cycles of treatment. The number of cycles depends on the situation but may be up to 12. Each treatment cycle lasts 2 weeks.
On the first day oxaliplatin is given through a drip over 2 hours. Folinic acid is given concurrently. This followed by an injection of fluorouracil into the cannula or central line. An infusion of 5FU is then started through a drip or pump, which lasts for 22 hours.
On the second day folinic acid is given as an injection or through a drip for 2 hours. This is followed by an injection of fluorouracil, followed by another fluorouracil infusion through a drip or pump for 22 hours.
Patient then has no treatment for 12 days. After that they start another treatment cycle.
If they have a central line they may be able to have the infusions of fluorouracil at home. If they are at home, they have the infusions through a small pump. They can keep the pump in a small bag, or a bag on a belt (like a bum bag). They’ll need to go back to the hospital for the second day of their treatment, to have the pump changed. Sometimes a chemotherapy nurse may be able to change the infusion at their home. In some countries (like Germany), it is common practice that patients disconnect the infusion pump themselves at home.
When the second infusion of 5FU is finished, the nurse will disconnect the drip and take the cannula out. If they have a central line this will stay in but it will be blocked with a plastic cap until they start your next treatment cycle.
Филиалы, дочерние клиники, отделения
Филиалы
- Хеликс на Гражданском
- Хеликс на Апраксином
- Хеликс в Волхове
- Хеликс в Выборге
- Хеликс в Красном Селе
- Хеликс в Никольском
- Хеликс в Стрельне
- Хеликс на Варшавской
- Хеликс на 9-й линии В.О.
- Хеликс на Социалистической
- Хеликс во Всеволожске
- Хеликс на Бабушкина 81к1
- Хеликс на Кременчугской
- Хеликс Большевиков 30
- Хеликс на Бабушкина 14
- Хеликс на Новочеркасском
- Хеликс на Пулковской
- Хеликс на Тазаева
- Хеликс на 2-м Муринском
- Хеликс в Кингисеппе
- Хеликс на Зайцева
- Хеликс на Коллонтай 17
- Хеликс на Коломяжском
- Хеликс в Колпино
- Хеликс в Кронштадте
- Хеликс на Туристской
- Хеликс на Еленинской
- Хеликс на Белы Куна
- Хеликс на Школьной
- Хеликс на Космонавтов
- Хеликс на Авиаконструкторов
- Хеликс на Бухарестской
- Хеликс на Ветеранов
- Хеликс на Каменноостровском
- Хеликс на Карповке
- Хеликс на Композиторов
- Хеликс на Королева
- Хеликс на Ленинском
- Хеликс на Лесной
- Хеликс на Маршала Захарова
- Хеликс на Московском
- Хеликс на Народной
- Хеликс на Наставников
- Хеликс на Науки
- Хеликс на Невском
- Хеликс на Новаторов
- Хеликс на Обуховской обороны
- Хеликс на Парашютной
- Хеликс на Площади Мужества
- Хеликс на Сампсониевском
- Хеликс на Среднем проспекте
- Хеликс на Уточкина
- Хеликс на Новоколомяжском
- Хеликс на Есенина
- Хеликс на Коллонтай 28
- Хеликс Отрадное
- Хеликс на Среднеохтинском
- Хеликс на Ораниенбаумской
- Хеликс на Кораблестроителей
- Хеликс в Пушкине
- Хеликс на Энтузиастов
- Хеликс Рыбацкое
- Хеликс на Художников
- Хеликс на Молодцова
- Хеликс на Токарева
- Хеликс в Сосновом Боре
- Хеликс на 5-й советской
- Хеликс в Тихвине
- Хеликс на Скобелевском
- Хеликс на Ветеранов 114
- Хеликс на Славы
- Хеликс на Фурштатской
- Хеликс на Железнодорожной
- Хеликс на Черной Речки
- Хеликс на Дачном
- Хеликс на Рихарда Зорге
- Хеликс на Камышовой
- Хеликс на Яхтенной
Дочерние клиники
- Хеликс, Диагностический центр и лабораторная служба (филиал на Луначарского)
- ДЦ Хеликс Кантемировский, медицинский семейный центр
- Хеликс, Диагностический центр и лабораторная служба (филиал в Купчино)
- Хеликс на Брянцева
- Хеликс, Диагностический центр (филиал “Балтийская жемчужина”)
- Хеликс на Лиговском, диагностический центр и лабораторная служба
- Хеликс, Диагностический центр и лабораторная служба (филиал на площади Победы)
- Хеликс на Руставели
- Хеликс, Диагностический центр и лабораторная служба (филиал на Техноложке)
- Хеликс, Диагностический центр лабораторной службы (филиал Полюстровский)
- Хеликс, Диагностический центр и лабораторная служба (филиал Тамбасовский)
- Хеликс, Диагностический центр (ДЦ Славянка)
- Хеликс, Диагностический центр и лабораторная служба (филиал в Автово)
- Хеликс на Парнасе, диагностический центр и лабораторная служба
Пациенты и критерии включения в исследование
Для включения в исследование пациенты должны были иметь возраст 18–75 лет и гистологически подтвержденную IV стадию колоректальной аденокарциномы (в соответствии с UICC). В этот анализ включались лишь пациенты, у которых ретроспективно было установлено наличие мутации гена KRAS в кодоне 12 или 13. Другими ключевыми критериями включения были балл общего состояния 2 или меньше в соответствии с ECOG, ожидаемая продолжительность жизни больше 3 месяцев, адекватная функция органов и наличие по крайней мере одного патологического образования, которое возможно было измерить. Критериями исключения были хирургическое вмешательство или лучевая терапия в пределах 6 недель до включения в исследование, перенесенное лечение ингибиторами топоизомеразы-1, анти-СЭФР препаратами или анти-РЭФР препаратами, а также перенесенное цитотоксическое лечение колоректального рака (за исключением адъювантной химиотерапии ≥6 месяцев до рандомизации).
Common complications6
More than 10 in every 100 people have one or more of the side effects listed below.
- An increased risk of getting an infection from a drop in white blood cells – it is harder to fight infections and you can become very ill. You may have headaches, aching muscles, a cough, a sore throat, pain passing urine, or you may feel cold and shivery. If you have a severe infection this can be life-threatening. Contact your treatment centre straight away if you have any of these effects or if your temperature goes above 38 °C. You will have regular blood tests to check your blood cell levels
- Tiredness and breathlessness due to a drop in red blood cells (anaemia) – you may need a blood transfusion
- Bruising more easily due to a drop in platelets – you may have nosebleeds or bleeding gums after brushing your teeth. Or you may have lots of tiny red spots or bruises on your arms or legs (known as petechia)
- Tiredness and weakness (fatigue) during and after treatment – most people find their energy levels are back to normal within 6 months to a year
- Numbness or tingling in the fingers and toes happens to nearly everyone having oxaliplatin and is usually worse if you are cold. You may have trouble with fiddly tasks such as doing up buttons. This can start a few days or weeks after treatment and usually goes away within a few months of the treatment finishing
- Feeling of nausea happens to about 7 out of every 10 people (70%) but is usually well controlled with anti-nausea drugs. If the nausea is not controlled, let your nurse know as there are other anti-nausea medicines that may work better for you
- Pain in the vein during the infusion of oxaliplatin or folinic acid – tell your nurse if this happens. Sometimes the drugs may need to be given more slowly
- Some people develop soreness, redness and peeling on the palms of the hands and soles of the feet (plantar palmar syndrome). This may cause tingling, numbness, pain and dryness
- Diarrhea happens to 6 out of 10 people (60%) – tell your doctor or nurse if it becomes severe, if you can’t drink to replace the lost fluid, or if it carries on for more than 3 days.Your nurse may give you anti diarrhea medicine to take home with you after chemotherapy
- A sore mouth (stomatitis) happens to 4 out of 10 people (40%)
- Women may stop having periods (amenorrhoea) but this may be temporary
- Loss of fertility – you may not be able to become pregnant or father a child after this treatment. Talk to your doctor before starting treatment if you think you may want to have a baby in the future. Men may be able to store sperm before starting treatment
FOLFOX4
Adjuvant treatment in patients with stage III colon cancer is recommended for 12 cycles, every 2 weeks. The recommended dose schedule given every two weeks is as follows:
Day 1: Oxaliplatin 85 mg/m2 IV infusion in 250-500 mL D5W and leucovorin 200 mg/m2 IV infusion in D5W both given over 120 minutes at the same time in separate bags using a Y-line, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
Day 2: Leucovorin 200 mg/m2 IV infusion over 120 minutes, followed by 5-FU 400 mg/m2 IV bolus given over 2–4 minutes, followed by 5-FU 600 mg/m2 IV infusion in 500 mL D5W (recommended) as a 22-hour continuous infusion.
FOLFOX4 regime
| FOLFOX4 | ||||
|---|---|---|---|---|
| drug | dose | administration | time | term |
| Oxaliplatin | 85 mg/m2 | IV infusion | 2 h | day 1 |
| Folinic acid | 200 mg/m2 | IV infusion | 2 h | day 1 + 2 |
| Fluorouracil | 400 mg/m2 | IV bolus | 2 min | day 1 + 2 |
| Fluorouracil | 600 mg/m2 | IV infusion | 22 h | day 1 + 2 |
Premedication with antiemetics, including 5-HT3 blockers with or without dexamethasone, is recommended.
Эффективность лечения
Показатель общей частоты ответа опухоли в популяции, получившей лечение в соответствии с протоколом, составил 44% (95% ДИ от 29% до 59%) в группе FOLFIRI плюс цетуксимаб и 48% (95% ДИ от 33% до 62%) в группе FOLFIRI плюс бевацизумаб (таблица 2). Показатель частоты контроля заболевания (ЧКЗ) достиг 90% (95% ДИ от 81% до 99%) в группе А по сравнению с 89% (95% ДИ от 80% до 98%) в группе В. Медиана времени до возникновения ремиссии была короткой в обеих ветвях лечения (ветвь А: 2,8 мес. по сравнению с ветвью В: 2,9 мес.).
Таблица 2. Данные относительно ответа опухоли и выживания
| Данные относительно выживания (ПНЛ: N = 96) | FOLFIRI + цетуксимаб (N = 50) | FOLFIRI + бевацизумаб (N = 46) | ра | ||
|---|---|---|---|---|---|
| Месяцы (95% ДИ) | Месяцы (95% ДИ) | ||||
| Медиана выживания без прогрессирования | 7,5 (5,7–10,4) | 8,9 (7,3–11,4) | 0,86 | ||
| Медиана общего выживания | 22,7 (18,3–27,0) | 18,7 (13,0–24,4) | 0,69 | ||
| Время до прогрессирования | 9,7 (7,4–11,2) | 8,9 (7,3–11,3) | 0,81 | ||
| Время до провала лечения | 5,1 (3,3–6,6) | 6,3 (5,4–7,2) | 0,39 | ||
| Время до ремиссии | 2,8 (1,9–3,0) | 2,9 (2,5–3,3) | 0,48 | ||
| Продолжительность ремиссии | 7,1 (6,0–11,4) | 8,6 (5,2–10,2) | 0,87 | ||
| Данные относительно ответа опухоли (ПОЭ: N = 87) | % | N | % | N | pb |
| Общая частота ответа опухолис % (95% ДИ) | 44 (29–59) | 18 | 48 (33–63) | 22 | 0,83 |
| Частота контроля заболеванияd % (95% ДИ) | 90 (81–99) | 37 | 89 (80–98) | 41 | 1,0 |
| Полный ответ | 2 | 1 | 2 | 1 | 1,0 |
| Частичный ответ | 42 | 17 | 46 | 21 | 0,83 |
| Стабильное заболевание | 46 | 19 | 41 | 19 | 0,67 |
| Прогрессирующее заболевание | 10 | 4 | 11 | 5 | 1,0 |
аЛог-ранк тест. bТест точности Фишера (двусторонний). сПолный плюс частичный ответ. dПолный плюс частичный ответ плюс стабильное заболевание. ПОЭ — популяция, которую оценивали относительно эффективности (получали лечение до первой оценки опухоли); ДИ — доверительный интервал; FOLFIRI — инфузионные 5-фторурацил, фолиновая кислота, иринотекан; ПНЛ — популяция, которую намеревались лечить. |
На момент проведения анализа возникли 93% событий, связанных с показателем ВБП, и 80% событий, связанных с показателем ОВ. Медиана выживания без прогрессирования составила 7,5 месяца в ветви А по сравнению с 8,9 месяца в ветви В . Медиана общего выживания составила 22,7 месяца в ветви А и 18,7 месяца в ветви В (СР = 0,86) (рис. 2).
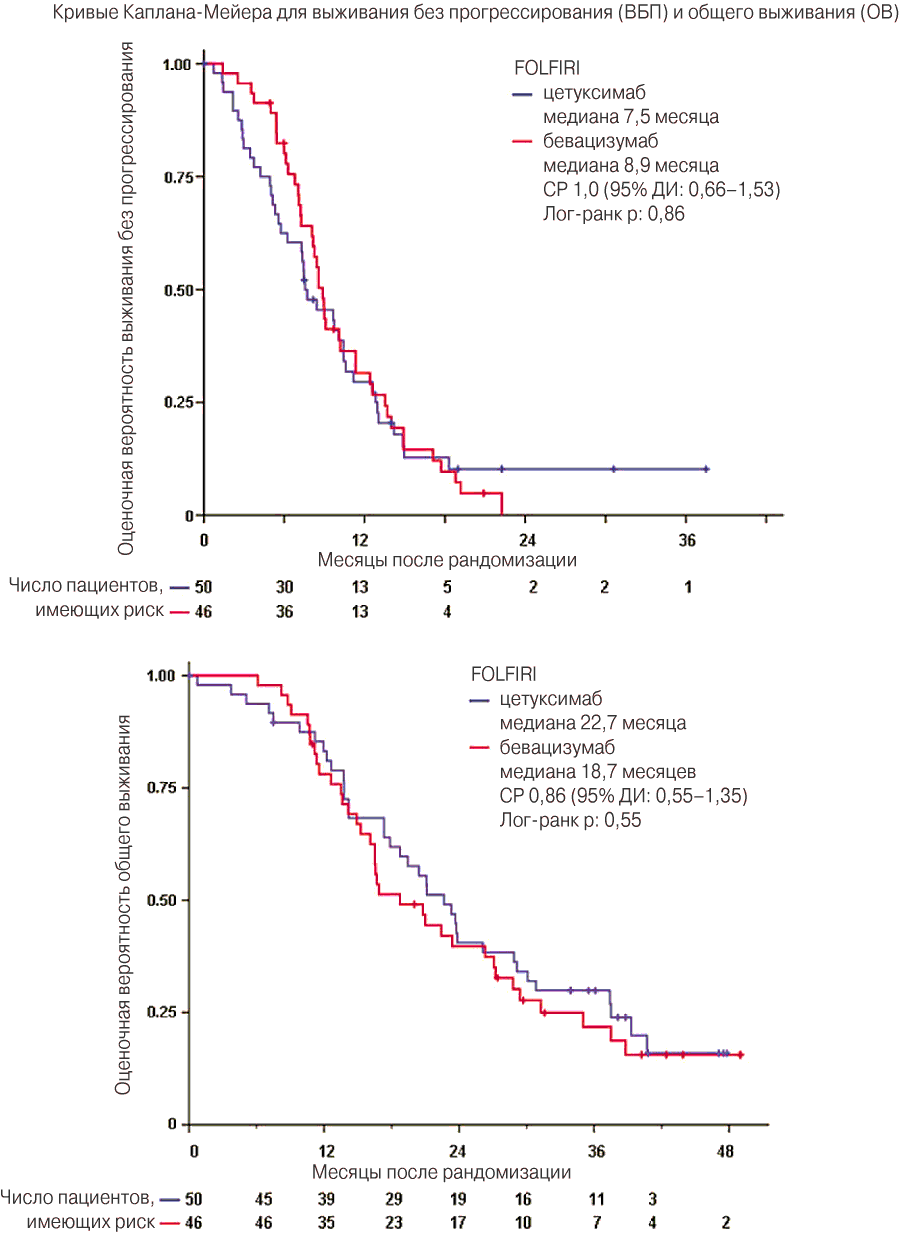
Рис. 2. Кривые Каплана-Мейера для выживания без прогрессирования и общего выживания.

Комментарии
(0 Комментариев)